 If you took Merida as directed and suffered Merida side effects, call Merida lawyer Timothy L. Miles today If you took Merida as directed and suffered Merida side effects, call Merida lawyer Timothy L. Miles today Meridia, made by Abbott Laboratories, was approved in 1997 for weight loss and weight loss maintenance for obese patients, and also very obese individuals who already had other health risk factors for cardiovascular disease. Meridia was approved after clinical studies showed that more patients taking Meridia lost at least 5 percent of their body weight than members of the placebo group who relied solely on diet and exercise. However, the U.S. Food & Drug Administration (FDA) has now requested that Meridia be recalled following a nearly year-long safety review of the drug which was initiated after researchers published the results of a study known as SCOUT (Sibutramine Cardiovascular Outcomes Trial). The study, which prompted a Meridia recall in Europe, showed a 16% percent increase in the risk of serious Meridia side effects, including:
SCOUT monitored 10,000 obese patients over the age of 55 who had a history of heart disease, and type 2 diabetes or similar risk factors. The initial purpose of the study was to show that weight loss through Meridia use lowered the risk of heart problems. However, that was not the case. In fact, the results suggested that those taking the weight loss drug had an increased chance of suffering Meridia cardiovascular side effects. Doctors have been advised to stop prescribing Meridia and begin discussing weight loss alternatives with their patients. WHAT IS MERIDIA? If you or a loved one suffered serious Meridia side effects, contact Meridia lawyer Timothy L. Miles today If you or a loved one suffered serious Meridia side effects, contact Meridia lawyer Timothy L. Miles today Meridia is a medication which requires a prescription and is used to treat the symptoms of obesity, weight loss, and maintenance of weight loss. It may be used alone or with other medications. Meridia belongs to a class of drugs called Schedule IV controlled substances and can cause severe Meridia side effects in some people. If you or a loved one suffered severe Meridia side effects call Meridia Lawyer Timothy L. Miles for a free case evaluation to see if you are eligible for a Meridia lawsuit and possibly entitled to substantial damages. IS MERIDIA STILL ON THE MARKET?Because of the seriousness of the Meridia side effects including the risk of a heart attack or stroke, Abbott agreed to voluntarily withdraw Meridia from the U.S. market because of clinical trial data indicating an increased risk of heart attack and stroke, according to the FDA. The FDA requested the market withdrawal after reviewing data from the Sibutramine Cardiovascular Outcomes Trial (SCOUT). SCOUT was initiated as part of a postmarket requirement to look at cardiovascular safety of sibutramine after the European approval of this drug. The trial demonstrated a 16 percent increase in the risk of serious heart events, including non-fatal heart attack, non-fatal stroke, the need to be resuscitated once the heart stopped, and death, in a group of patients given sibutramine compared to another given placebo. There was a small difference in weight loss between the placebo group and the group that received sibutramine. what are the SERIOS MERIDIA SIDE EFFECTSIn the beginning, Meridia seemed to be a booming success as it was used by millions of individuals around the world. However, hundreds of users began reporting serious Meridia side effects including:
Patients taking Meridia have also experienced:
Additionally, approximately 30 deaths were caused by Merida in the United States. Researchers looked at 10,000 patients who were obese, over the age of 55 with a history of risk factors and found that 11.4% of patients using Meridia suffered heart problems, compared to 10% given a sugar pill. As of June 30, 2009, the FDA Adverse Event Reactions (AERS) database indicates that at least 84 Meridia deaths from cardiovascular causes have been reported to the FDA.' If you suffered severe Meridia side effects call Meridia Lawyer Timothy L. Miles for a free case evaluation to see if you are eligible for a Meridia lawsuit. Was MERIDIA RECALLED AFTER SERIOUS MERIDIA SIDE EFFECTS REPORTEDThe limited benefits provided by Meridia are outweighed by the increased risk of cardiovascular problems which prompted federal regulators to issue the recall for Meridia in October 2010. Lawyers are investigating and considering potential lawsuits against Abbott on behalf of individuals who suffered a:
If you took Meridia as directed and suffered serious Meridia side effects including a heart attack or stroke, then you may meet the requirements for a Merida lawsuit and possibly be entitled to substantial compensation. Contact Merida Lawyer Timothy L. Miles today to see if you are eligible for a Merida lawsuit. SHOULD I REPORT MERIDIA SIDE EFFECTS?Yes, the FDA urges consumers who experience adverse advents to any medication or product to report it via the MedWatch Online Voluntary Reporting Form. When reporting your Merida side effects make sure to be through and give as many details as possible about your experience taking the drug including all Merida side effects you suffered, and any other symptoms you suffered that you believe was caused by Merida use. IF YOU SUFFERED MERIDIA SIDE EFFECTS, CALL A MERIDIA LAWYER TODAYIf you or a loved one suffered serious Meridia side effects including a heart attack, stroke or even death, contact Meridia lawyer Timothy L. Miles today to see if you are eligible for a Meridia lawsuit and possibly entitled to substantial compensation. While there is still time to file a lawsuit, be mindful that these cases are time sensitive, and give us a call today and see if you meet the requirements for a Meridia lawsuit. timothy l. miles, esq.Timothy L. Miles is a nationally recognized shareholder and consumer rights attorney raised in Nashville, Tennessee. Mr. Miles was recently selected by Martindale-Hubbell® and ALM as a 2023 Top Ranked Lawyer, 2023 Top Rated Litigator. and a 2023 Elite Lawyer of the South. Mr. Miles also maintains the AV Preeminent Rating by Martindale-Hubbell®, their highest rating for both legal ability and ethics. Mr. Miles is a member of the prestigious Top 100 Civil Plaintiff Trial Lawyers: The National Trial Lawyers Association, a superb rated attorney by Avvo, a recipient of the Lifetime Achievement Award by Premier Lawyers of America (2019) and recognized as a Distinguished Lawyer, Recognizing Excellence in Securities Law, by Lawyers of Distinction (2019). Mr. Miles has published over sixty articles on various issues of the law, including class actions, whistleblower cases, products liability, and more.  If you suffered serious side effects from a prescription drug, call Timothy L. Miles today If you suffered serious side effects from a prescription drug, call Timothy L. Miles today Side effects are any unwanted or unexpected effects after taking a prescription drug. While all drugs come with side effects, most are minor and are significantly outweighed by the drug’s benefits. Unfortunately, in some cases, in a rush to market, some drugs can have devastating side effects causing major health consequences, including death. For example, in the Belviq class action lawsuit, Belviq was a popular prescription weight-loss drug until a U.S. Food and Drug Administration (FDA) safety trial concluded that the drug could increase the risk of several different types of cancer, including pancreatic, lung, and colorectal cancer. As a result, the FDA issued a full Belviq recall in February 2020 and as a result, numerous Belviq lawsuits have been filed including a Belviq class action lawsuit. Similarly, in the Meridia lawsuit, involving another weight loss drug, Meridia was taken off the market because of the seriousness of the Meridia side effects including the risk of a heart attack or stroke. TYPES OF SIDE EFFECTSThe most common side effects people report to taking prescription drugs include:
However, more serios side effects that could be fatal include:
For example, in the Xeljanz lawsuits it is allege people suffered serious Xeljanz Side Effects including blood clots, in the lungs, heart attacks, strokes, breast and colon cancer, and other severe health problems. The Actemra lawsuit alleges the drug for rheumatoid arthritis cases serios Acatemra side effects including heart attacks, strokes, heart failure, and interstitial lung disease. The Tasigna lawsuit alleges a prescription medication used to treat chronic myeloid leukaemia causes serious Tasigna side effects including heart or circulation problems (including complications like blocked arteries, heart attacks, or strokes). A possible side effect for all pregnant women taking prescription drugs is having a baby with birth defects. Therefore, Pregnant women should always speak to their health care provider about any prescription drugs or supplements before taking them. THE FDA’S FUNCTION RELATED TO SIDE EFFECTSA drug must be approved by the FDA before it can be put on the market. Any New Drug Applications (NDAs) submitted by pharmaceutical companies must provide evidence that the drug has its intended effect and is safe. Evidence comes from testing the drug, first in animals and then in humans. Once the drug is determined safe and efficient, it will be approved by the FDA if it deems that its benefits outweigh its risks. Nevertheless, in some instances a drug’s side effects will not show up until after the medication enters the marketplace and more people start using it. MedWatch is the FDA’s post-marketing surveillance program which seeks voluntary input, mainly from health care professionals, on unwanted effects they see every day. If these reports are numerous or serious enough, the FDA will take regulatory action such as requiring the manufacturer to add warnings to a drug’s label. Additionally, The FDA also wants input from consumers. Prescription drugs must have s label that contains a toll-free number the agency has for letting it know about side effects with prescription drugs, referred to as "adverse events." Consumers can report severe side effects by call MedWatch at 1-800-FDA-1088 or by reporting serious problems to the FDA on its website. Sometimes, the FDA approved the new drug but have certain reservations about the drug and order the manufacture to perform a post-marketing study. If the post-marketing study comes back with new or severe side effects it will take action. For example, in both the Kombiglyze XR Lawsuit and the Onglyza Lawsuit, the FDA ordered safety warning be pot on all Onglyza and Kombiglyze XR bottles that the drugs could result in cardiovascular injury after the manufacturers’ post-marketing study revealed the cardiovascular side effects, among others. If you suffered Kombiglyze XR side effects or Onglyza side effects, you may be eligible for compensation. Contact class action and products liability Timothy L. Miles today for a free case evaluation. CONCLUSIONMost prescription drug side effects are minor and wear off over time. However, some side effects can be very dangerous, even life threatening. If you suffered Actemra side effects, Tasigna side effects, Kombiglyze XR side effects or Onglyza side effects or any serious side effect from any prescription drug, contact Nashville attorney Timothy L. Miles today. TIMOTHY L. MILES, ESQ.imothy L. Miles is a nationally recognized shareholder and consumer rights attorney raised in Nashville, Tennessee. Mr. Miles was recently selected by Martindale-Hubbell® and ALM as a 2023 Top Ranked Lawyer, 2023 Top Rated Litigator. and a 2023 Elite Lawyer of the South. Mr. Miles also maintains the AV Preeminent Rating by Martindale-Hubbell®, their highest rating for both legal ability and ethics. Mr. Miles is a member of the prestigious Top 100 Civil Plaintiff Trial Lawyers: The National Trial Lawyers Association, a superb rated attorney by Avvo, a recipient of the Lifetime Achievement Award by Premier Lawyers of America (2019) and recognized as a Distinguished Lawyer, Recognizing Excellence in Securities Law, by Lawyers of Distinction (2019). Mr. Miles has published over sixty articles on various issues of the law, including class actions, whistleblower cases, products liability, and more.  If you suffered serios side effects from a prescription drug call Timothy L. Miles If you suffered serios side effects from a prescription drug call Timothy L. Miles Recently, there have been numerous product and drug recalls by the U.S. Food and Drug Administration (FDA) in the news prompting numerous lawsuits including the Philips CPAP lawsuit, the Belviq Class Action Lawsuit, the Valsartan Lawsuit, the Meridia lawsuit and many others. When a product is recalled by the FDA, this means the drug or product has been determined to be unsafe or potentially dangerous as was the case in the Meridia Lawsuit. It is vital to understand the FDA recall process and why it is necessary to protect the public. The FDA recall process is a very complex system that includes a numerous steps and stages that must be taken in order to ensure the safety of consumers. It begins with the FDA issuing a recall notice for the affected product, and then involves a variety of steps to ensure that the product is removed from the market. The recall process also includes the manufacturer taking specific steps to address the safety issue and investigate the cause of the recall. Understanding the recall process can help consumers to make informed decisions and stay safe from potential hazards. WHAT DOES IT MEAN WHEN A PRODUCT IS RECALLED?A product recall is a removal of a product that has been deemed unsafe and potentially dangerous to consumers and is in violation of laws administered by the FDA. A majority of the time, a product recall occurs after a company has discovered that the product is defective or contains a health risk as happened in the Philips CPAP lawsuit. The company might have also received reports of injuries or deaths related to the product. The FDA issued a recall notice and will require the products be removed from the market, as was the case in the Belviq Class Action Lawsuit because of serious Belviq side effects including headache, dizziness and nausea. The company may issue a recall voluntarily before the FDA contacts them or issues a recall notice as in the Meridia Lawsuit involving serious Meridia side effects including a heart attack, stroke, resuscitated cardiac arrest or even death, A product recall can occur in a variety of ways. The most common way that a product is recalled is through a customer or consumer complaint. If a consumer finds a problem with a product, such as a broken piece, or if it fails to function properly, they may report the issue to the company. If the company receives enough complaints about a specific product, they may decide to issue a recall. The company may also decide to initiate a recall after being notified by the FDA about a safety issue or receiving a warning letter from the agency. WHAT IS THE FDA RECALL PROCESS? If you were injured from a recalled prescription drug, call Timothy L. Miles If you were injured from a recalled prescription drug, call Timothy L. Miles The FDA recall process has several stages that may occur simultaneously or gradually over time. It begins with a company discovering a problem with a product or receiving a complaint from a customer. The company will then decide whether to initiate a voluntary recall or wait to see if the FDA issues a recall notice. If the FDA detects a problem with a product, they may issue a recall notice. A notice will be issued if the company fails to act on a complaint or if there is a serious risk to the public that requires immediate action.
HOW DOES THE FDA DECIDE WHEN TO ISSUE A RECALL?The FDA recall process begins as soon company becomes aware of a problem with a product. Sometimes, the company may decide to issue a voluntary recall such as the recalled Philips CPAP Machines. The company will determine if the issue is serious enough to warrant a recall and if they are able to repair the product. The FDA will issue a recall notice if they detect a serious risk to the public. The FDA may also issue a recall notice if a company fails to act on a complaint or take the necessary actions to address a problem with a product. There are several factors that may be considered when deciding if a recall is necessary. The type of product and the defect may be considered. The size of the company and the resources available for a recall may also be considered. The FDA will also consider the potential harm that the product can cause to the public. The potential for the product to cause harm will be weighed against the potential for serious harm if the product is recalled. WHAT ARE THE STAGES OF THE FDA RECALL PROCESS?There are several stages that make up the FDA recall process. These include notification, voluntary recall, and mandatory recall.
RECALL CLASSIFICATIONSThe FDA classifies recalls as either a Class I, II or III in order to indicate the relative degree of health hazard presented by the product being recalled.
HOW DOES THE RECALL PROCESS PROTECT CONSUMERS?The FDA recall process is used to remove unsafe or potentially dangerous products from the market that may cause serious health risks. The process protects consumers by removing the products and helping them avoid injury or death. The FDA recall process also helps to maintain consumer confidence in the products that are available to purchase. Consumers can protect themselves by reading product recalls and instructions carefully. It is important to follow the instructions given on labels and instructions so that the product will function properly and be safe to use. Consumers should report any potential defects or issues with products to the manufacturer. Taking these steps can help to ensure that defective products are removed from the market and that the public remains safe. WHAT ARE THE RESPONSIBILITIES OF THE MANUFACTURER DURING THE RECALL PROCESS?The company that manufactures the product that is recalled has several responsibilities during the recall process. The company must issue a recall notice and inform the public of the recall. The company must also take the product off the shelf and stop selling the product. Manufacturers will also take specific actions to address the defect with the product. The company may offer repair or replacement services if they are able to fix the product efficiently as is the case in the Philips CPAP lawsuit. If a company fails to take the necessary steps to address a recall, the FDA may issue a mandatory recall. The company will be responsible for paying all costs associated with the recall. This may include finances used to issue the recall notice, public relations costs, and expenses related to removing the product from the shelf. The FDA may also impose fines on a company for failing to recall a product in a timely manner. WHAT STEPS CAN CONSUMERS TAKE TO STAY AWARE OF PRODUCT RECALLS?Consumers can stay aware of product recalls by staying informed and reading product labels and instructions carefully. Consumers can also sign up for recall notifications through the FDA website. These notifications will be sent if a product is being recalled. It is important for consumers to report any potential defects or issues with products to the manufacturer and MedWatch, the FDAs reporting program for health professionals, patients and consumers. Taking these steps can help to ensure that defective products are removed from the market and that the public remains safe. CONTACT THE LAW OFFICES OF TIMOTHY L. MILESIf you were injured or suffered serious side effects including serious health issues from a recalled drug or product, contact Timothy L. Miles, a Nashville and top rated class action lawyer today for a free case evaluation. You may be entitled to significant compensation, and we are here to help. TIMOTHY L. MILES, ESQ.Timothy L. Miles is a nationally recognized shareholder and consumer rights attorney raised in Nashville, Tennessee. Mr. Miles was recently selected by Martindale-Hubbell® and ALM as a 2023 Top Ranked Lawyer, 2023 Top Rated Litigator. and a 2023 Elite Lawyer of the South. Mr. Miles also maintains the AV Preeminent Rating by Martindale-Hubbell®, their highest rating for both legal ability and ethics. Mr. Miles is a member of the prestigious Top 100 Civil Plaintiff Trial Lawyers: The National Trial Lawyers Association, a superb rated attorney by Avvo, a recipient of the Lifetime Achievement Award by Premier Lawyers of America (2019) and recognized as a Distinguished Lawyer, Recognizing Excellence in Securities Law, by Lawyers of Distinction (2019). Mr. Miles has published over sixty articles on various issues of the law, including class actions, whistleblower cases, products liability, and more.
Call Elmiron Lawyer Timothy L. Miles if you suffered Elmiron Eye Damage
Since 1996, millions of patients have been prescribed Elmiron in order to treat their interstitial cystitis (IC), otherwise known as painful bladder syndrome. But there is now reason to believe that this drug may cause a serious eye condition in those taking it, one that may result in blindness.
This condition is a form of maculopathy – the degeneration of the macula, the central part of the retina. Maculopathy’s first symptoms are blurred vision, dark spots, trouble focusing, and other impairments: as it progresses, it may lead to total blindness. As the evidence has accumulated, the manufacturer of Elmiron – Janssen Pharmaceuticals – has failed to warn the public about these risks. Millions of people are taking a drug that may one day rob them of their vision, and an untold number may have already suffered serious health conditions. Elmiron eye damage lawsuits on behalf of the injured have already begun claiming their maculopathy was caused by Elmiron. If you or a loved one took Elmiron and has since been diagnosed with maculopathy, you may be eligible for compensation. Call an Elmiron Lawyer today to have your case reviewed for free and see if you qualify for the Elmiron eye damage lawsuit. A New Danger Comes to Light
The potential risks associated with Elmiron were unknown until 2015, when doctors at Atlanta’s Emory Eye Center encountered an eye condition they had notice seen before in six women. This form of maculopathy didn’t seem to have a cause, but further investigation revealed that all six women had been under long-term use of Elmiron for years.
Further studies released in 2018 and 2019 further support the link that maculopathy is caused by Elmiron. Although, it is currently unknown how maculopathy is caused by Elmiron. Is maculopathy caused by Long-Term Use of Elmiron?
In 2018, researchers at the Emory Eye Center at the Emory School of Medicine in Atlanta alerted the public that taking Elmiron is linked to the retinal condition, maculopathy. Pentosan polysulfate sodium can cause severe vision problems and side effects, including interstitial cytitis, and even blindness.
Nieraj Jain, M.D., of Emory Eye Center in Atlanta, Ga., reported that six Elmiron patients who had been taking Elmiron for about 15 years had developed unusual changes in their macula. Because nothing in the patients’ medical history or diagnostic tests explained the subtle, but striking pattern of abnormalities, Dr. Jain and his colleagues raised a warning flag that long-term use of Elmiron may damage the retina. The retina is a thin layer of tissue that lines the back of the eye on the inside. It is located near the optic nerve. The retina receives light that the lens has focused and converts the light into neural signals. It then sends these signals on to the brain for visual recognition. All patients in the study reportedly showed signs of retinal disease and that the youngest patient in the study was diagnosed with interstitial cystitis at 23 years of age. That patient began taking the medication daily and began showing visual symptoms by the age of 30. In addition, three ophthalmologists conducted a review of patients at Kaiser Permanente in Northern California and found that approximately one-quarter of patients with significant exposure to Elmiron showed definite signs of eye damage. It also found the medication could potentially be linked to retinal conditions, such as age-related macular degeneration or pattern dystrophy. Patients who have used or are using the pentosan polysulfate sodium drug Elmiron are advised to be screened by ophthalmologists if they are experiencing any unusual eye symptoms. They are also advised to cease pentosan therapy if eye disease is present. What Is the Criteria for Filing An Elmiron Lawsuit?
The current criteria for filing an Elmiron lawsuit requires:
How Much Are Elmiron Settlements?
The litigation against the pharmaceutical company is new so there are no Elmiron settlements yet. For most drug manufacturer lawsuits, the cases can take several years before there are any compensation payout amount determinations. When it gets to that stage of the litigation process, there is a specific claim submission process. Every Elmiron lawsuit claimant will submit a claim detailing their injuries and side effects and they are supported by the medical records. The claimants with more serious injuries will receive higher settlements. The loss of vision or decrease in vision is life-altering for the patient and family. It is expected these eye damage lawsuits to result in substantial settlement payouts.
Call an Elmiron Lawyer about an Elmiron Eye Damage Lawsuit
If you or a loved one took Elmiron and have been diagnosed with maculopathy caused by Elmiron, you could be owed compensation for medical costs, pain and suffering, lost wages, and other damages. Call Elmiron Lawyer Timothy L. Miles today and see what he can do for you.
Timothy L. MILES, ESQ.Timothy L. Miles is a nationally recognized shareholder and consumer rights attorney raised in Nashville, Tennessee. Mr. Miles was recently selected by Martindale-Hubbell® and ALM as a 2023 Top Ranked Lawyer, 2023 Top Rated Litigator. and a 2023 Elite Lawyer of the South. Mr. Miles also maintains the AV Preeminent Rating by Martindale-Hubbell®, their highest rating for both legal ability and ethics. Mr. Miles is a member of the prestigious Top 100 Civil Plaintiff Trial Lawyers: The National Trial Lawyers Association, a superb rated attorney by Avvo, a recipient of the Lifetime Achievement Award by Premier Lawyers of America (2019) and recognized as a Distinguished Lawyer, Recognizing Excellence in Securities Law, by Lawyers of Distinction (2019). Mr. Miles has published over sixty articles on various issues of the law, including class actions, whistleblower cases, products liability, and more.
Contact Onglyza lawyer Timothy L. Miles today to see if you are eligible for a Onglyza lawsuit
Onglyza (saxagliptin) and Kombiglyze XR (saxagliptin and metformin) belong to a new class of diabetic drugs known as DPP-4 inhibitors. Onglyza gained federal drug approval by the U.S. Food & Drug Administration (FDA) despite initial concerns over the cardiovascular safety of the drug.
These concerns were echoed in a post-marketing safety study that found Onglyza may be associated with an increased risk of heart failure and an increased risk of death. If you or a loved one took Onglyza as directed and suffered from Onglyza side effects including heart failure or other cardiovascular injuries—such as congestive heart failure or myocardial infarction, after starting Onglyza therapy, contact Onglyza lawyer Timothy L. Miles today to see if you are eligible for a Onglyza lawsuit and possibly entitled to substantial compensation. WHAT IS ONGLYZA?
Onglyza (saxagliptin) is an add-on medication to diet and exercise to improve blood sugar control in people with type 2 diabetes. It is a is a dipeptidyl peptidase-4 (DPP-4) inhibitor that increases the amount of insulin your body makes. Onglyza comes in tablets in dosages of 2.5 mg and 5 mg taken once by mouth daily.
WHO SHOULD NOT TAKE ONGLYZA?
You should not take Onglyza if you are allergic to any of its ingredients. Serious allergic reactions can occur with Onglyza including swelling of the face, throat, lips and difficulty breathing, among others. Your doctor should test your blood to measure how well your kidneys work before and during treatment with Onglyza. You may need a lower dose of Onglyza if your kidneys are not working well.
WHAT IS THE PROBLEM WITH ONGLYZA? Call an Onglyza lawyer if you suffered serious Onglyza side effects Call an Onglyza lawyer if you suffered serious Onglyza side effects
Type 2 diabetes patients have a significantly increased risk of cardiovascular complications, and the majority of diabetics die from such complications. Amid growing concerns over the safety of many diabetes drugs, in 2008 the FDA recommended that the makers of new type 2 diabetes drugs provide evidence that the drugs do not increase the risk of cardiovascular events such as heart attacks.
Onglyza met the FDA's cardiovascular safety criteria. Though the FDA approved the new diabetic drug, it had reservations about the sufficiency of Onglyza clinical trials and ordered AstraZeneca to perform a post-marketing study. As a result, AstraZeneca completed a 16,000-patient cardiovascular outcomes trial called SAVOR. The results of that trial were published in the New England Journal of Medicine and raised major Onglyza safety concerns. SAVOR found that Onglyza patients were 27% more likely to be hospitalized for heart failure. The study also found a significant increase in the rate of “all-cause mortality” among Onglyza patients. The warning signs of heart failure include:
Following the release of SAVOR, the FDA issued an Onglyza drug safety communication and convened an advisory committee meeting to analyze the results and recommend further action. The committee voted 14-to-1 to add information to Onglyza’s label warning about heart failure risks. The other member voted to withdraw Onglyza from the U.S. market. The committee, however, did not advise updating the warning label to address the measured increase in all-cause mortality among Onglyza patients and did not recommend any restrictions on prescribing the drug. In an April 2016 drug safety communication, the FDA announced that Onglyza and Kombiglyze XR are now required to carry warnings about heart failure risk. Other FDA warnings about saxagliptin include:
What Are the Onglyza Side Effects?
In addition to heart failure, Onglyza is linked to these adverse side effects:
HOW DO I KNOW IF I MEET THE REQUIREMENTS FOR AN ONGLYZA LAWSUIT?
If you or a loved one took Onglyza as directed and suffered suffered heart failure, death, or another serious cardiovascular injury, after starting Onglyza therapy, you may meet the requirements for a Onglyza lawsuit and possibly be entitled to substantial compensation. Call a Onglyza lawyer for free today who can tell you if you are eligible for a Onglyza lawsuit.
CONTACT AN ONGLYZA LAWYER TO SEE IF YOU ARE ELIGIBLE FOR AN ONGLYZA LAWSUIT?
If you or a loved one took Onglyza as directed and suffered from Onglyza Side Effects, specifically: heart failure, death or another serious cardiovascular injury —such as congestive heart failure or myocardial infarction, after starting Onglyza therapy, contact Onglyza lawyer Timothy L. Miles today to see if you are eligible for a Onglyza lawsuit and possibly entitled to substantial compensation.
timothy l. miles, esq.Timothy L. Miles is a nationally recognized shareholder and consumer rights attorney raised in Nashville, Tennessee. Mr. Miles was recently selected by Martindale-Hubbell® and ALM as a 2023 Top Ranked Lawyer, 2023 Top Rated Litigator. and a 2023 Elite Lawyer of the South. Mr. Miles also maintains the AV Preeminent Rating by Martindale-Hubbell®, their highest rating for both legal ability and ethics. Mr. Miles is a member of the prestigious Top 100 Civil Plaintiff Trial Lawyers: The National Trial Lawyers Association, a superb rated attorney by Avvo, a recipient of the Lifetime Achievement Award by Premier Lawyers of America (2019) and recognized as a Distinguished Lawyer, Recognizing Excellence in Securities Law, by Lawyers of Distinction (2019). Mr. Miles has published over sixty articles on various issues of the law, including class actions, whistleblower cases, products liability, and more.
If you or a loved one suffered serious Actemra side effects while taking Actemra, contact Actemra lawyer Timothy L Miles
Roche’s Actemra was heralded as a breakthrough therapy for rheumatoid arthritis when it was introduced in 2010.
The drug’s website proclaims that “Actemra works differently” and shows a patient happily scuba diving and walking on a tropical beach. But nowhere does Roche mention certain side effects that may have contributed to more than 1,100 patient deaths. Research suggests that Actemra patients, compared to those taking competing rheumatoid arthritis drugs, face similar risks for heart attacks, heart failure, strokes, lung disease, and pancreatitis. But unlike the competition, Actemra’s label does not warn about these side effects. If you or a loved one suffered serious Actemra side effects including heart, lung, or pancreatic side effects while taking Actemra, contact Actemra lawyer Timothy L. Miles today to see if you are eligible for an Actemra lawsuit. If you meet the requirements for an Actemra lawsuit you may be entitled to substantial compensation. WHAT IS ACTEMRA? Call an Actemra lawyer today if you suffered serious Actemra side effects Call an Actemra lawyer today if you suffered serious Actemra side effects
Actemra (tocilizumab) is a medication currently approved to treat adults with moderately to severely active rheumatoid arthritis (RA). Actemra blocks the inflammatory protein IL-6, which improves joint pain and swelling from arthritis and other symptoms caused by inflammation. You must have tried and failed another medication for RA before starting Actemra.
Actemra is given as an injection once a week or as an IV infusion once a month. If you are on the infusion, you will have this medicine at a center where nurses will place a needle in your vein and give you the medicine through it. It takes about one hour for the medicine to be infused. Actemra is given every 4 weeks. If you are on the injection, you will be taught to give yourself a needle. Actemra comes in a pre-filled syringe. WHAT ARE THE ACTEMRA SIDE EFFECTS TO THE HEART AND LUNGS?
A STAT News report and postmarketing studies raise questions about the adequacy of Actemra's safety warnings.
STAT reviewed more than 500,000 adverse event reports on leading rheumatoid arthritis drugs, including Actemra, Humira, and Remicade. STAT found evidence that Roche downplayed Actemra’s cardiovascular, pancreatic, and lung side effects. According to STAT, from 2010 to 2016 the U.S. Food and Drug Administration (FDA) logged more than 13,500 Actemra adverse event reports, including 1,128 patients who died after taking Actemra. While safety data indicate the risks of heart attacks, strokes, heart failure, and interstitial lung disease are as high or higher for Actemra patients than for patients taking some similar drugs, Actemra’s warning label—unlike its competition's—does not reflect these risks. For example, Actemra patients reported more cases of interstitial lung disease than Remicade patients, and nearly the same number as Humira patients. But while Remicade and Humira have interstitial lung disease warnings, Actemra does not. STAT’s findings are supported by postmarketing studies—clinical studies conducted after a pharmaceutical product goes on the market to better understand drug risks. The FDA ordered Actemra postmarketing studies because premarket testing revealed potentially worrisome side effects. One such study compared Actemra to another arthritis drug, Enbrel, and found rates of stroke and heart failure were about 1.5 times higher in Actemra users. Enbrel has strong warnings for patients with cardiovascular disease. Actemra does not have similar warnings despite similar or worse cardiovascular risks. The takeaway from these findings is that doctors and patients may not be getting a complete picture of Actemra’s risk-benefit profile. DRUG MANUFACTURERS HAVE A DUTY TO WARN
Pharmaceutical products are not expected to be completely without harmful side effects. But if a drugmaker understands that a drug presents a certain risk, it has a duty to put an appropriate warning label on the product. That way patients and their doctors can evaluate all known risks and benefits and make an educated decision about whether a medication is right for them.
While the FDA has authority to demand warning labels, the responsibility ultimately falls on drug manufacturers. Actemra has garnered FDA attention since it was approved in 2010. At one point the agency considered a pancreatitis warning label. But Genetech/Roche talked the FDA out of the warning. The agency then approved broader use of Actemra for giant cell arteritis. Conflicts of interest between manufacturers and regulators plague the drug safety system. The STAT report details how all 11 authors of an Actemra postmarketing study had financial ties to Roche or Genetech. And a former FDA manager who oversaw Actemra’s approval for rheumatoid arthritis now works for Roche, where he helps gain FDA approval for new uses of the drug. The drug safety system, in other words, cannot always be trusted to put patient safety first. That’s why, when patients suffer harm from drug side effects, they often must turn to the legal system. How Do I Know if I am Eligible for an Actemra Lawsuit?
We are looking into claims that Genetech/Roche failed to warn about the following Actemra side effects:
Actemra patients who suffered any of these side effects may be eligible for an Actemra lawsuit. IF I MEET THE REQUIREMENTS FOR AN ACTEMRA LAWSUIT, WHAT CAN I GET?
If you meet the requirements for an Actemra lawsuit and are successful you can recover money for some of the following damages:
• Past and future medical bills (including medication, hospital stays, and in-home care) • Pain and suffering • Lost wages • Loss of earning capacity • Funeral expenses (in the event of a loved one’s death) Broadly speaking, a plaintiff who is eligible for an Actemra lawsuit could be entitled to compensation for any past and future costs associated with their diagnosis if they meet the requirements for an Actemra lawsuit and are successful. CALL AN ACTEMRA LAWYER TODAY IF YOU SUFFERED SERIOUS ACTEMRA SIDE EFFECTS?
If you or a loved one suffered serious Actemra side effects including heart, lung, or pancreatic side effects while taking Actemra, contact Actemra lawyer Timothy L Miles today to see if you are eligible for an Actemra lawsuit. If you meet the requirements for an Actemra lawsuit you may be entitled to substantial compensation.
timothy l. miles, esq.Timothy L. Miles is a nationally recognized shareholder and consumer rights attorney raised in Nashville, Tennessee. Mr. Miles was recently selected by Martindale-Hubbell® and ALM as a 2023 Top Ranked Lawyer, 2023 Top Rated Litigator. and a 2023 Elite Lawyer of the South. Mr. Miles also maintains the AV Preeminent Rating by Martindale-Hubbell®, their highest rating for both legal ability and ethics. Mr. Miles is a member of the prestigious Top 100 Civil Plaintiff Trial Lawyers: The National Trial Lawyers Association, a superb rated attorney by Avvo, a recipient of the Lifetime Achievement Award by Premier Lawyers of America (2019) and recognized as a Distinguished Lawyer, Recognizing Excellence in Securities Law, by Lawyers of Distinction (2019). Mr. Miles has published over sixty articles on various issues of the law, including class actions, whistleblower cases, products liability, and more.
Contact a Xeljanz Lawyer today and see if you meet the Xeljanz lawsuit criteria if you suffered Xeljanz side effects
What should you do when a medication makes you feel worse, not better? Whether it’s over-the-counter medicine or a prescription from a doctor, drugs are meant to treat a patient. However, when the side effects exceed the benefits, medicines can become very dangerous and sometimes lethal.
Pharmaceutical companies and suppliers have both an ethical and legal responsibility to properly disclose the risks and side effects of a drug before bringing it to market, but all too often those warnings are withheld in the name of profit. However, in just the last two years, the U.S. Food and Drug Administration (FDA) has warned that Xeljanz (Tofacitinib) can be associated with heart problems, blood clots, and even cancer. But the question remains: why didn’t Pfizer warn consumers about these potential risks sooner? Xeljanz lawsuits claim that Pfizer failed to give adequate warning about the Xeljanz side effects, which caused preventable harm to patients who suffered blood clots in the lungs, heart attacks, strokes, breast and colon cancer, and other severe health problems. We are accepting these cases on a contingency-fee basis. That means our clients don’t pay unless we win. If you need to discuss a lawsuit with a Xeljanz lawyer, free of charge, please contact us. (855) Tim-M-Law (855-846-6529) or [email protected] ABOUT XELJANZ If you or a loved one suffered Xeljanx side effects, contact a Xeljanz lawyer today If you or a loved one suffered Xeljanx side effects, contact a Xeljanz lawyer today
Xeljanz belongs to a class of drugs known as Janus kinase (JAK) inhibitors. As Pfizer explains, our body has signaling proteins called cytokines that regulate immune system responses. The production of cytokines is increased when our body fights infection. Cytokine production is thought to play a role in chronic inflammatory conditions, such as rheumatoid arthritis, psoriatic arthritis, and ulcerative colitis. Xeljanz disrupts signaling that produces some of the cytokines, reducing inflammation. Specifically, Xeljanz blocks—or inhibits—the release of cytokines in the JAK signal pathway.
Xeljanz has been approved for several different uses:
Xeljanz has been approved for several different uses:
POST-MARKETING XELJANZ STUDY REVEALS SERIOUS SAFETY CONCERNS
Most Americans assume that if a drug is approved by the FDA, it has been thoroughly evaluated for safety and efficacy. However, this isn’t always the case. In addition to pre-approval clinical testing, the FDA may order post-marketing studies as a condition of drug approval. These trials gather more information about a drug’s safety, efficacy, or optimal use. Here, the Xeljanz study reveals serious safety concerns.
When the FDA first approved Xeljanz in 2012, it ordered a post-marketing Xeljanz safety study known as ORAL Surveillance. But rather than confirming the safety of Xeljanz, the post-marketing Xeljanz Study Revealed Serious Safety Concerns and has led to new health alerts and warnings. SOME PATIENTS TOOK XELJANZ AND DEVELOPED BLOOD CLOTS
In February 2019, the FDA warned that the ongoing Xeljanz safety trial found an increased risk of taking Xeljanz and developing blood clots in the lungs and death. In July 2019, the FDA announced that it had approved new warnings about thrombosis and death in patients treated with a 10 mg twice-daily dose of Xeljanz.
Thrombosis occurs when a blood clot blocks a vein or artery. Deep vein thrombosis, pulmonary embolism, and arterial thrombosis have all been reported in Xeljanz patients. A blood clot in a deep vein is known as a deep vein thrombosis (DVT). DVT usually occurs in the legs. A blood clot that travels to the lungs is called a pulmonary embolism (PE). PE is usually caused by a blood clot that starts in the arm or the leg. Arterial thrombosis is a blood clot in an artery. This type of thrombosis can stop blood from reaching vital organs. Blood clots can lead to a heart attack, stroke, organ damage, and even death. Symptoms of a blood clot include:
XELJANZ AND CANCER
The next major safety news to come out of the Xeljanz post-marketing study was equally concerning because of Xeljanz and Cancer. The FDA alerted the public in February 2021 that preliminary results showed an increased risk of cancer with Xeljanz.
Pfizer reports in a press release that malignancies observed in Xeljanz studies include (but are not limited to):
Also in February 2021, the FDA warned about serious heart-related problems that may result from Xeljanz use. Pfizer refers to these in its press release as “major adverse cardiovascular events” (MACE). Pfizer reports that a total of 135 patients in the study had a major adverse cardiovascular event, although the full results have not yet been released. The most frequently reported event was myocardial infarction (i.e. heart attack).
We are currently speaking to clients across the country about Xeljanz cancer, blood clot, and cardiovascular lawsuits. Patients who took Xeljanz as prescribed and developed a malignancy, thrombosis, or heart-related issues may qualify for a lawsuit. Lawsuits against Pfizer can recover compensation for medical bills, lost wages, pain and suffering, reduced quality of life, and more. XELJANZ AND MAJOR CARDIOVASCULAR EVENTS
The latest results from the ORAL Surveillance study were released in early 2021 and led to an FDA safety communication about increased risk of serious heart-related problems, or “major adverse cardiovascular events” (MACE).
Pfizer reported 135 patient subjects with MACE. The most frequently reported such event was myocardial infarction (heart attack), but the full data has not yet been released. Other major cardiac events that may be linked to Xeljanz include:
SHOULD I STOP TAKING XELJANZ?
Anyone who is currently using Xeljanz should speak with their doctor about terminating their prescription to avoid the dangerous Xeljanz side effects. Not only will they help you stop the drug, but they can also advise you on safe medication alternatives for your condition.
HOW DO I KNOW IF I MEET THE XELJANZ LAWSUIT CRITERIA?
If you took Xeljanz and have been diagnosed with cancer, a major adverse cardiovascular event, or venous thromboembolism (such as a pulmonary embolism or deep vein thrombosis) or other serious Xeljanz side effects, you may qualify for a Xeljanz lawsuit and be eligible for substantial compensation. Call today for a free case evaluation and see if you meet the Xeljanz Lawsuit Criteria.
CONTACT A XELJANZ LAWYER ABOUT A XELJANZ LAWSUIT
Xeljanz, while marketed as a safe medication, increases the risk of blood clots, cancer, and cardiovascular problems for its users. The FDA approved the drug upon release, but a post-marketing safety study revealed serious safety concerns and concluded that the side effects are more detrimental than initially believed. The company responsible for the medication, Pfizer, knew about the side effects prior to the safety study but failed to disclose this information to the public in an attempt to retain their profits. This unethical behavior exposed thousands of consumers to an elevated risk of harm, making their actions unacceptable by any means.
Unfortunately, some users of Xeljanz have developed cancer as a result of their medication use, including but not limited to lymphoma, melanoma, and lung cancer. Pfizer, already knowing the risk of the Xeljanz side effects, sold it to consumers anyway, causing many innocent individuals to develop and suffer from a life-threatening disease. Their reluctance to disclose the risk of their product is not just unethical, it’s illegal, and our attorneys are here to help hold them accountable to the full extent of the law. Contact a Xeljanz Lawyer today at (855) 846-6529 or [email protected] and see if you meet the Xeljanz lawsuit criteria if you suffered Xeljanz side effects. Someone will promptly get in touch with you. We are here to help! TIMOTHY L. MILES, ESQ.Timothy L. Miles is a nationally recognized shareholder and consumer rights attorney raised in Nashville, Tennessee. Mr. Miles was recently selected by Martindale-Hubbell® and ALM as a 2023 Top Ranked Lawyer, 2023 Top Rated Litigator. and a 2023 Elite Lawyer of the South. Mr. Miles also maintains the AV Preeminent Rating by Martindale-Hubbell®, their highest rating for both legal ability and ethics. Mr. Miles is a member of the prestigious Top 100 Civil Plaintiff Trial Lawyers: The National Trial Lawyers Association, a superb rated attorney by Avvo, a recipient of the Lifetime Achievement Award by Premier Lawyers of America (2019) and recognized as a Distinguished Lawyer, Recognizing Excellence in Securities Law, by Lawyers of Distinction (2019). Mr. Miles has published over sixty articles on various issues of the law, including class actions, whistleblower cases, products liability, and more.
If you suffered Valsartan Side Effects, contact Valsartan lawyer Timothy L. Miles today
Patients who have taken Valsartan and have been diagnosed with cancer, as a result, should read on to learn more about how they can hold the drug makers accountable.
Valsartan has been recalled due to possible nitrosamine contamination. Nitrosamines can cause tumors in the liver and other organs in lab animals and are thought to be carcinogenic in humans as well. It is believed that these blood pressure drugs were tainted during flawed manufacturing processes. The U.S. Food and Drug Administration (FDA) stated that these contaminants “are of special concern to global regulators because, unlike most impurities in drugs, they have the potential to cause harm at very low levels.” The recalls began in July when the FDA found that some Valsartan products contained a potentially cancer-causing chemical. European regulators had already made the same discovery. The substance can form during manufacturing if the chemical reactions used to make the drug are not carefully controlled and monitored, the FDA said. Valsartan is made by Chinese manufacturer Zhejiang Huahai Pharmaceuticals. These recalls are a product of greater FDA scrutiny of foreign drug imports, but FDA warnings alone will not ensure safe medications. In a culture where the profit motive exists, drug companies must be held accountable to cutting corners and sacrificing consumer safety. If you or a loved one took Valsartan and later were diagnosed with cancer or other serious Valsartan Side Effects, contact Valsartan lawyer Timothy L. Miles today to see if you meet the Valsartan lawsuit criteria and may be entitled to substantial compensation. What Prompted the Valsartan Recall?
In July 2018, the FDA announced the Valsartan Recall due to contamination. The recall was prompted because of the discovery of a cancer-causing agent in the drug. The FDA launched an investigation into the cause and impact of the contamination which led to several additional recalls over the following year. The FDA also found that other blood pressure drugs contained impurities.
The first recall was prompted by the finding of the impurity N-nitrosodimethylamine (NDMA). Later, the FDA later discovered the second impurity,N-Nitrosodiethylamine (NDEA). While neither compound has been studied in humans, both have been shown to cause cancer in animal studies. What Is Valsartan?
Valsartan belongs to a class of drugs called angiotensin II receptor blockers (ARB). Valsartan is a blood pressure medication used to treat high blood pressure and heart failure. It is also prescribed after a heart attack to lower blood pressure and help the patient live a longer life. The medication should not be taken while pregnant because it may be harmful to an unborn child.
What are the Valsartan Side Effects?
In addition to cancer, sources say other Valsartan Side Effects include:
Have Lawsuits been filed over Possible Valsartan Side Effects?
Yes, patients who filed Valsartan Cancer Lawsuits were prescribed Valsartan claim that drug manufacturers willfully disregarded required safety procedures at the plant where contamination occurred. The contaminant was undiscovered for years until revealed by random testing.
How Do I Know if I Meet the Valsartan Lawsuit Criteria?
If you took Valsartan and subsequently were diagnosed with cancer, or experienced other serious Valsartan side effects, then you may meet the Valsartan lawsuit criteria and may be eligible for substantial compensation. Contact Valsartan Lawyer Timothy L. Miles today and see if are eligible for a Valsartan lawsuit.
Call a Valsartan Lawyer about a Valsartan Lawsuit
If you or a loved one have received a cancer diagnosis or experienced other serious Valsartan side effects after taking Valsartan, contact us for a free, no-risk case evaluation to see if you meet the Valsartan lawsuit criteria. We might be able to help you pursue compensation for the harm you and your loved ones have suffered if you meet the Valsartan lawsuit criteria.
Call a Valsartan Lawyer today to see how he can help you. A Valsartan Lawyer can explain the process and answer any questions you may have including whether you meet the Valsartan lawsuit criteria. TIMOTHY L. MILES, ESQ.Timothy L. Miles is a nationally recognized shareholder and consumer rights attorney raised in Nashville, Tennessee. Mr. Miles was recently selected by Martindale-Hubbell® and ALM as a 2023 Top Ranked Lawyer, 2023 Top Rated Litigator. and a 2023 Elite Lawyer of the South. Mr. Miles also maintains the AV Preeminent Rating by Martindale-Hubbell®, their highest rating for both legal ability and ethics. Mr. Miles is a member of the prestigious Top 100 Civil Plaintiff Trial Lawyers: The National Trial Lawyers Association, a superb rated attorney by Avvo, a recipient of the Lifetime Achievement Award by Premier Lawyers of America (2019) and recognized as a Distinguished Lawyer, Recognizing Excellence in Securities Law, by Lawyers of Distinction (2019). Mr. Miles has published over sixty articles on various issues of the law, including class actions, whistleblower cases, products liability, and more.
More than 7,000 people have filed a Byetta lawsuit against the makers of the drug Byetta since its release in 2003. The most common Byetta lawsuits stem from the drug’s dangerous side effects, which can include hepatitis and pancreatic cancer, as well as claims that Pfizer and AstraZeneca withheld information about the drug’s risks and potential side effects. If you or someone you know took Byetta and were diagnosed with pancreatic cancer, contact Byetta lawsuit lawyer Timothy L. Miles to see if you are eligible for a Byetta lawsuit.
If you took Byetta and were diagnosed with pancreatic cancer, contact Byetta lawsuit lawyer Timothy L. Miles
Byetta Background
The drug Byetta was developed and released in 2003. It contains the ingredients Exenatide and human insulin, and it comes in the form of a once-daily injection. While the drug was approved for the treatment of Type 2 diabetes, it is also used for obesity. According to the FDA, Byetta is indicated as an "adjunct to diet and exercise for the treatment of type 2 diabetes mellitus in patients inadequately controlled on diet and exercise." The drug is also indicated for "combination therapy in patients with type 2 diabetes mellitus who require initial combination therapy." It is also indicated for “adjunctive therapy to diet and exercise for the prevention of diabetic complications.” Byetta is manufactured and distributed by Pfizer and AstraZeneca.
Byetta Side Effects
Byetta side effects include, among others:
Pfizer and Byetta Lawsuits Contact a Byetta lawsuit lawyer today if you took Byetta and were diagnosed with pancreatic cancer Contact a Byetta lawsuit lawyer today if you took Byetta and were diagnosed with pancreatic cancer
Pfizer and AstraZeneca are being sued by people who suffered serious side effects as a result of using Byetta. The side effects include pancreatitis, pancreatic cancer, and other serious injuries. In March and April 2019, two federal class action lawsuits were filed against Pfizer and AstraZeneca regarding Byetta. The lawsuits were filed in the U.S. District Court for the Southern District of New York by litigants on behalf of all individuals who were harmed by the drug. The lawsuits allege that Pfizer and AstraZeneca engaged in deceptive marketing when they aggressively marketed Byetta to doctors and patients. The lawsuits also allege that Pfizer and AstraZeneca failed to inform doctors of the risks associated with the drug.
AstraZeneca and Byetta Lawsuits
In addition to being sued, AstraZeneca has been named in many Byetta lawsuits as a co-defendant. The lawsuits allege that the company failed to provide adequate warnings about the risks and side effects of the drug. They also claim that the company failed to monitor the use and adverse events associated with the drug. The first Byetta lawsuit against AstraZeneca was filed in November 2018. The lawsuit was filed by a man who used Byetta and allegedly suffered a heart attack as a result of the drug. The lawsuit claimed that the drug’s warning label was inadequate and failed to disclose the risk associated with the use of the drug.
Dangerous Ingredients in Byetta
Some of the ingredients in Byetta have been linked to serious side effects. These include Exenatide and human insulin. Exenatide, one of the ingredients in Byetta, is associated with an increased risk of pancreatitis, a serious condition that can result in hospitalization and death. The insulin that makes up another part of Byetta has been associated with hypersensitivity and allergic reactions, including anaphylaxis.
Conclusion
The drug Byetta has been linked to a wide range of serious side effects. It has also been linked to an increased risk of cancer in some patients. If you or a loved one took Byetta and were diagnosed with pancreatic cancer, you may be eligible for a Byetta lawsuit. Contact Byetta lawsuit lawyer Timothy L. Miles today and see if you meet the requirements for a Byetta lawsuit and could be eligible for substantial compensation. While there is still time to file a lawsuit, be mindful that these cases are time sensitive, and give us a call today.
TIMOTHY L. MILES, ESQ.Timothy L. Miles is a nationally recognized shareholder and consumer rights attorney raised in Nashville, Tennessee. Mr. Miles was recently selected by Martindale-Hubbell® and ALM as a 2023 Top Ranked Lawyer, 2023 Top Rated Litigator. and a 2023 Elite Lawyer of the South. Mr. Miles also maintains the AV Preeminent Rating by Martindale-Hubbell®, their highest rating for both legal ability and ethics. Mr. Miles is a member of the prestigious Top 100 Civil Plaintiff Trial Lawyers: The National Trial Lawyers Association, a superb rated attorney by Avvo, a recipient of the Lifetime Achievement Award by Premier Lawyers of America (2019) and recognized as a Distinguished Lawyer, Recognizing Excellence in Securities Law, by Lawyers of Distinction (2019). Mr. Miles has published over sixty articles on various issues of the law, including class actions, whistleblower cases, products liability, and more.
If you experienced Zostavaz side effects contact Zostavaz lawyer Timothy L. Miles today
Zostavax is a vaccine approved by the Food and Drug Administration (FDA) in 2016 as a way to reduce the risk of shingles and its complications in adults aged 60 and older. However, soon after Zostavax was released to the public, there were numerous reports of Zostavax side effects that appeared to be directly linked to its use. Now, several Zostavax lawsuits have been filed against the manufacturer. If you or a loved one suffered serious Zostavax side effects including shingles, blindness or even death, contact Zostavax lawyer Timothy L. Miles today to see if you are eligible for a Zostavax lawsuit and possibly entitled to substantial compensation.
What is Zostavax?
Zostavax became the first shingles vaccine to be approved by the U.S. Food and Drug Administration (FDA) in 2006. FDA. Zostavax is administered as a shot to the upper and works using a live, but weakened form, of the varicella-zoster virus to decrease the risk of shingles. The varicella-zoster virus also causes chickenpox in children.
It was thought to be highly effective at the time and Zostavax seemed to be an important breakthrough in the containment of the disease. Zostavax would help Merck earn nearly $750 million in profits over the next 12 years. Who Is Eligible to File a Zostavax Lawsuit?
Individuals of all ages who have experienced severe or debilitating Zostavax side effects as a result of receiving Zostavax may be eligible to file a Zostavax lawsuit and receive compensation for damages related to the vaccine. Zostavax is approved for people who are at least 50 years of age, but some people in their 40s have also been prescribed it.
When deciding whether to apply for the vaccine, it is important to weigh the benefits against the potential side effects and risks. Not everyone who is at risk is a good candidate for the vaccine. Because Zostavax is a live vaccine, it is not appropriate for people who are immunocompromised or pregnant. Most of the Zostavax lawsuits focus on individuals who have experienced serious Zostavax side effects. If you or a loved one suffered serious Zostavax side effects including shingles, blindness or even death, contact us today. (855) Tim-M-Law (855-846-6529) Product Liability and Defects in Manufacturing
Zostavax lawsuits often highlight the fact that the FDA’s warning label for the vaccine reads that “Zostavax has not been proven to prevent shingles in people 50 years of age or older,” meaning that the vaccine’s manufacturer cannot guarantee that it will prevent shingles in any age group. In addition, some Zostavax lawsuits point to the vaccine’s problematic manufacturing process as an indication of product liability.
This manufacturing process may have included the use of human fetal cells, which is a known source of contamination linked to the development of infectious diseases. While the FDA did approve Zostavax in 2016, it is important to note that they did so with the recommendation that the manufacturer conducts more research on the vaccine’s effectiveness in adults 50 years of age and older. Whether or not the manufacturer failed to conduct this follow-up research, the FDA failed to hold them accountable for it, or both remains unclear as the Zostavax lawsuit proceeds. Negligence and Failure to Warn
Some Zostavax lawsuits include claims of negligence on the part of the manufacturer, suggesting that the company failed to inform patients about the true risks of the vaccine or failed to provide adequate warning about its side effects. In addition to these allegations of negligence, some lawsuits also include allegations of failure to warn, which suggest that the manufacturer knew of the vaccine’s side effects but failed to warn patients about them. While some vaccine injury lawsuits are filed on behalf of individuals who were injured or contracted an illness related to the vaccine, others are filed on behalf of individuals who experienced no negative side effects but want to raise awareness about the potential risks associated with the vaccine.
Conclusion
If you or a loved one suffered serious Zostavax side effects including shingles, blindness or even death, contact Zostavax lawyer Timothy L. Miles today to see if you meet the requirements for a Zostavax lawsuit and possibly may be entitled to substantial compensation. While there is still time to file a lawsuit, be mindful that these cases are time sensitive, and give us a call today. (855) Tim-M-Law (855-846-6529).
TIMOTHY L. MILESTimothy L. Miles is a nationally recognized shareholder and consumer rights attorney raised in Nashville, Tennessee. Mr. Miles was recently selected by Martindale-Hubbell® and ALM as a 2023 Top Ranked Lawyer, 2023 Top Rated Litigator. and a 2023 Elite Lawyer of the South. Mr. Miles also maintains the AV Preeminent Rating by Martindale-Hubbell®, their highest rating for both legal ability and ethics. Mr. Miles is a member of the prestigious Top 100 Civil Plaintiff Trial Lawyers: The National Trial Lawyers Association, a superb rated attorney by Avvo, a recipient of the Lifetime Achievement Award by Premier Lawyers of America (2019) and recognized as a Distinguished Lawyer, Recognizing Excellence in Securities Law, by Lawyers of Distinction (2019). Mr. Miles has published over sixty articles on various issues of the law, including class actions, whistleblower cases, products liability, and more. 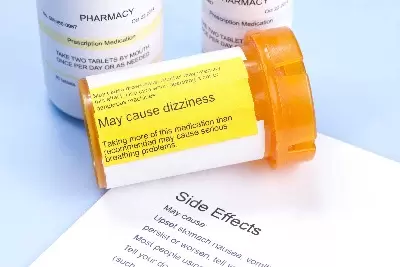 If you suffered Kombiglyze XR side effects, contact a Kombiglyze XR lawyer today If you suffered Kombiglyze XR side effects, contact a Kombiglyze XR lawyer today Kombiglyze XR is a combination of metformin (a biguanide) and saxagliptin (a DPP4 inhibitor). Kombiglyze XR works by lowering the amount of sugar made by your liver, decreasing the amount of sugar absorbed by your intestine, increasing the amount of insulin your body makes, and helps your body respond better to insulin. However, research shows that type 2 diabetes drug Kombiglyze XR (saxagliptin and metformin) may cause heart failure or other cardiovascular injuries in some patients. According to the Centers for Disease Control and Prevention (CDC), 30 million Americans have diabetes and 84 million have pre-diabetes. Metformin—one of the drugs found in combination drug Kombiglyze—is among the top 10 most popular prescription drugs taken by Americans. Analysts also predict that Onglyza (saxagliptin), Kombiglyze’s sister drug, could reach $1.8 billion in sales in 2018. In 2015, roughly 386,000 people were prescribed Onglyza or Kombiglyze XR, the U.S. Food and Drug Administration (FDA) reported. Given that so many people take these drugs, any side effects they may cause that are severe enough to hospitalize patients should be treated with concern. Dozens of patients are filing lawsuits against Kombiglyze’s manufacturer AstraZeneca and its previous partner Bristol-Myers Squibb alleging that the companies failed to adequately test the drugs’ safety prior to selling it, which ultimately caused their cardiovascular injuries. If you or a loved one took Kombiglyze XR as directed and suffered from Kombiglyze XR side effects including heart failure or other cardiovascular injuries—such as congestive heart failure or myocardial infarction, after starting Kombiglyze XR therapy, contact Kombiglyze XR lawyer Timothy L. Miles today to see if you are eligible for a Kombiglyze XR lawsuit and possibly entitled to substantial compensation. Read on for answers to five frequently asked questions about a Kombiglyze XR lawsuit. What Are the Kombiglyze XR Side Effects?The following are some of the common Kombiglyze XR side effects:
Additionally, dozens of lawsuits allege other Kombiglyze XR side effects including serious cardiovascular injury. What Is the Problem with Kombiglyze XR?Research shows Kombiglyze XR and other similar drugs may put patients at risk of heart complications. Dozens of patients are filing lawsuits against Kombiglyze’s manufacturer AstraZeneca and its previous partner Bristol-Myers Squibb alleging that the companies failed to adequately test the drugs’ safety prior to selling it, which ultimately caused their cardiovascular injuries. Does Kombiglyze XR Put Patients at Risk of Heart Failure?A clinical trial funded by AstraZeneca and Bristol-Myers Squibb was one of the first studies to report cardiovascular health risks associated with these drugs. Saxagliptin Assessment of Vascular Outcomes Recorded in Patients with Diabetes Mellitus, or the SAVOR trial, found that patients with a history of heart problems who took Onglyza or Kombiglyze XR had a 27% greater chance of being hospitalized for heart failure than patients who took a placebo. Patients with chronic kidney disease and / or previous heart failure were more likely to be hospitalized. When an FDA panel reviewed the results from the SAVOR study, 14 out of 15 panelists voted to require a safety warning on all Onglyza and Kombiglyze XR bottles that the drugs could result in cardiovascular injury. What Do the Kombiglyze XR Lawsuits Allege?Kombiglyze XR lawsuits allege that AstraZeneca and Bristol-Myers Squibb failed to:
If I Meet the Requirements for a Kombiglyze XR lawsuit, What Can I Get?If you meet the requirements for a Kombiglyze XR lawsuit and are successful you can recover money for some of the following damages: • Past and future medical bills (including medication, hospital stays, and in-home care) • Pain and suffering • Lost wages • Loss of earning capacity • Funeral expenses (in the event of a loved one’s death) Broadly speaking, a plaintiff who is eligible for a Kombiglyze XR lawsuit could be entitled to compensation for any past and future costs associated with their diagnosis if they meet the requirements for a Kombiglyze XR lawsuit and are successful. ConclusionIf you or a loved one took Kombiglyze XR as directed and suffered from Kombiglyze XR Side Effects, specifically: heart failure or other cardiovascular injuries—such as congestive heart failure or myocardial infarction, after starting Kombiglyze XR therapy, contact Kombiglyze XR lawyer Timothy L. Miles today to see if you are eligible for a Kombiglyze XR lawsuit and possibly entitled to substantial compensation. TIMOTHY L. MILESTimothy L. Miles is a nationally recognized shareholder rights attorney raised in Nashville, Tennessee. Mr. Miles was recently selected by Martindale-Hubbell® and ALM as a 2022 Top Ranked Lawyer, 2022 Top Rated Litigator. and a 2022 Elite Lawyer of the South. Mr. Miles also maintains the AV Preeminent Rating by Martindale-Hubbell®, their highest rating for both legal ability and ethics. Mr. Miles is a member of the prestigious Top 100 Civil Plaintiff Trial Lawyers: The National Trial Lawyers Association, a superb rated attorney by Avvo, a recipient of the Lifetime Achievement Award by Premier Lawyers of America (2019) and recognized as a Distinguished Lawyer, Recognizing Excellence in Securities Law, by Lawyers of Distinction (2019). Mr. Miles has published over sixty articles on various issues of the law, including class actions, whistleblower cases, products liability, and more. Please visit our website.  Call a Belviq lawyer today to see if you meet the criteria for a Belviq lawsuit Call a Belviq lawyer today to see if you meet the criteria for a Belviq lawsuit Belviq (generic name: lorcaserin) is a popular prescription weight-loss drug made by Eisai Inc. to help people experiencing obesity and weight-related health issues curb their hunger. While it is not known exactly how Belviq works, the drug targets a hunger receptor in the brain and helps people fee full after easting smaller amounts of food and also seems to help people fee full longer. Alarmingly, a U.S. Food and Drug Administration (FDA) safety trial concluded that the drug could increase the risk of several different types of cancer, including pancreatic, lung, and colorectal cancer. Those individuals who participated in the clinical trial reported certain types of cancer with some types occurring more frequently in the lorcaserin group, including pancreatic, colorectal, and lung. As a result, the FDA issued a full Belviq recall in February 2020. In response, numerous Belviq lawsuits have been filed including a Belviq class action. If you or a loved one took Belviq, Belviq XR, or another brand of lorcaserin, and you were diagnosed with cancer, or other serious Belviq side effects, you may be entitled to file a Belviq lawsuit or be included in a Belviq class action lawsuit and receive compensation from the drug’s manufacturer. Fill out a free case review form to find out if you are owed money for damages and talk to a Belviq Lawsuit Lawyer about whether you meet the Belviq Lawsuit Criteria and are eligible for a Belviq lawsuit or to be a part of the Belviq class action lawsuit today at no charge. Read on for answers to eight frequently asked questions about a Belviq Class action lawsuit. What Kind of Drug is Belviq?Belviq belongs to a class of drugs which includes Central Nervous System (CNS) Stimulants; Anorexiants; and Serotonin 5HT2C-Receptor Agonist. The drug is used by certain overweight people, such as those who are obese or have weight-related medical problems. Losing weight and keeping it off can lessen the many health risks that come with obesity, including heart disease, diabetes, high blood pressure, and a shorter life, among others. Belviq is thought to work by affecting a certain part of the brain that helps control your appetite. Belviq was available by prescription in its original form and in an extended-release version, Belviq XR. Both versions are the subject of Belviq lawsuits. Arena Pharmaceuticals manufactures Belviq at its facility in Switzerland. Eisai Inc. marketed and distributed Belviq in the United States. Both companies are named defendants in lawsuits. What Are the Belviq Side Effects?The most common Belviq side effects include headache, dizziness and nausea. People with diabetes also commonly suffered hypoglycemia, back pain and cough. Other common Belviq side effects include:
Does Belviq Cause Cancer?Weight-loss drugs are not known for their safety; unfortunately, Belviq is no exception resulting in a Belviq recall. In a five-year, double-blind study of 12,000 patients, those taking Belviq were more likely to be diagnosed with cancer than those taking a placebo (sugar pill). At the end of the five-years, the FDA found 7.1% of people in the placebo group developed cancer compared to 7.7% of people in the Belviq group who developed cancer. The FDA stated, “A range of cancer types was reported, with several different types of cancers occurring more frequently in the lorcaserin group,” including: Pancreatic cancer, colorectal cancer; and Lung cancer. What is the Risk in Taking Belviq?In February 2020, the FDA requested the Belviq Recall and for to Eisai pull all Belviq and Belviq XR products from the market due to Belviq Side Effects. Eisai has complied with this request. As a result, Belviq is no longer commercially available. Why Did the FDA Recall Belviq?When the FDA announced the Belviq Recall it stated: "We are taking this action because we believe that the risks of lorcaserin outweigh its benefits based on our completed review of results from a randomized clinical trial assessing safety." What Are the Legal Claims in a Belviq Lawsuit?While the attorneys are still researching and reviewing the facts to determine all the claims to bring in a Belviq Lawsuit, two claims that will undoubtably be brought in these lawsuits are a defective design claim and a claim for the failure to warn of the risks of cancer. With a design defect, a drug manufacturer can be held strictly liable if: (1) the drug's chemical makeup posed a foreseeable risk; and (2) the drug was designed and administered as it was intended to be. Evidence surfaced in early 2010 that Belviq caused cancer in rats and the FDA originally denied the drug's application for this reason. Therefore, the defendants should have known that the "chemical makeup" increased the odds for getting cancer. With a failure to warn claim drug manufacturers are under a duty to warn doctors and medical providers of the drug's non-obvious hazards and inform them of ways to avoid these dangers. Physicians who prescribed Belviq to patients had no idea of the cancer risks or other Belviq Side Effects. Had the defendant warned physicians they may have decided not to prescribe it to their patients and patients who knew of the risks may have decided to not take it. What Is the Criteria for a Belviq Lawsuit?If you took Belviq or its generic equivalent and were diagnosed with cancer, you should consider taking legal action against the drug’s manufacturer. If you meet these criteria, you may have a case and can file a belviq class action lawsuit. -Over the age of 18 -Diagnosed with cancer after taking Belviq, Belviq XR, or another brand of lorcaserin If a loved one took this weight loss drug and was diagnosed with cancer, you may also contact a Belviq Lawsuit Lawyer on their behalf about a Belviq class action lawsuit. Call Belviq Lawyer Timothy L. Miles today (855-Tim-M-Law) for a free and confidential case evaluation. What Can I Recover from a Belviq Lawsuit?Every case is unique, but people who successfully file drug or medical device lawsuits can recover money for some of the following damages: • Past and future medical bills (including medication, hospital stays, and in-home care) • Pain and suffering • Lost wages • Loss of earning capacity • Funeral expenses (in the event of a loved one’s death) Broadly speaking, a plaintiff in a Belviq lawsuit could be entitled to compensation for any past and future costs associated with their cancer diagnosis. Call a Belviq lawsuit lawyer about a Belviq LawsuitIf you took Belviq and have been diagnosed with cancer, you could be owed significant compensation for medical costs, pain and suffering, lost wages, and other damages. You should speak with Belviq lawyer Timothy L. Miles as soon as possible to determine the best course of action including filing a Belviq class action lawsuit or an individual Belviq Lawsuit if you were diagnosed with cancer or experienced other severe Belviq side effects. These cases are time-sensitive, including the Belviq class action lawsuit, so do not wait. Fill out a case review form to receive a free, no-obligation case evaluation. It costs nothing to get started, a Belviq lawyer can explain the process and answer any questions you may have. We take all cases on a contingency basis which means we do not get paid unless we win your case. Call today and see what a Belviq Lawyer can do for you. TIMOTHY L. MILESTimothy L. Miles is a nationally recognized shareholder and consumer rights attorney raised in Nashville, Tennessee. Mr. Miles was recently selected by Martindale-Hubbell® and ALM as a 2023 Top Ranked Lawyer, 2023 Top Rated Litigator. and a 2023 Elite Lawyer of the South. Mr. Miles also maintains the AV Preeminent Rating by Martindale-Hubbell®, their highest rating for both legal ability and ethics. Mr. Miles is a member of the prestigious Top 100 Civil Plaintiff Trial Lawyers: The National Trial Lawyers Association, a superb rated attorney by Avvo, a recipient of the Lifetime Achievement Award by Premier Lawyers of America (2019) and recognized as a Distinguished Lawyer, Recognizing Excellence in Securities Law, by Lawyers of Distinction (2019). Mr. Miles has published over sixty articles on various issues of the law, including class actions, whistleblower cases, products liability, and more. |
|
CONTACT
The Law Offices of Timothy L. Miles Tapestry at Brentwood Town Center 300 Centerview Dr., #247 Brentwood, TN 37027 Phone: (855) 846-6529 Email: [email protected] HOURS OF OPERATION Mon-Fri: 24/7 Sat-Sun: 24/7 |
CONNECt FacebooK
|
